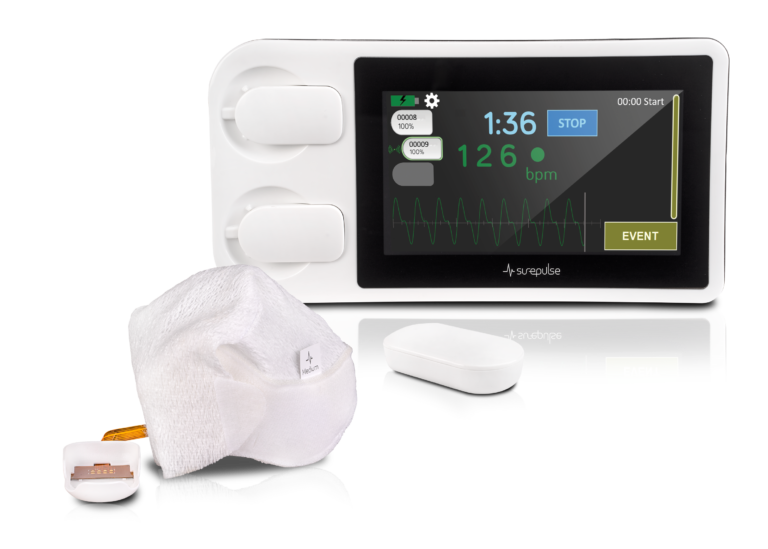
SurePulse Medical Ltd, a spin-out from MICRA partner the University of Nottingham, have developed a wireless heartrate monitor for new born babies. The SurePulse VS overcomes many problems that impact upon the effectiveness of traditional ways of monitoring the health and wellbeing of newborn babies needing additional support.
There is substantial need for more effective ways of monitoring the heartrate of new born babies. Every year 80,000 babies in the UK require some form of stabilisation or resuscitation at birth. This equates to around 10% of the country’s births. If this figure is scaled up across the entire world (140million births), each year 14million babies require stabilisation or resuscitation at the very start of their life.
For babies needing additional support at birth the heart rate is a primary indicator of well-being, and a significant body of medical research illustrates that some of the primary existing methods of monitoring newly born baby’s key vital signs possess practical drawbacks as well as significant scope for inaccuracies, and therefore potential patient harm.
Investors have recognised the scope and potential for SurePulse’s technology and products to improve neonatal healthcare and deliver returns on investment. In July 2020 it was announced that new and existing investors would be injecting a further £1million into to company to support its further growth.
As SurePulse’s growth continues we spoke to the company’s CEO Dr.James Carpenter about what it has taken to get this far and develop an innovative regulatory-approved device.
James’ background is in electrical engineering, having completed a PhD and undertaken postdoctoral research in medical device electronics before joining the SurePulse team and leading the spin-out.
He explains that SurePulse’s journey began with “…a meeting of minds between the faculties of Engineering and Medicine at the University of Nottingham. Engineers in the department of Electronic and Electrical Engineering were working on an optical vital sign monitoring technology for miners working in hostile environments. At an interdisciplinary meeting, an academic clinician heard the engineers talking about the project and enquired whether the technology might be suitable for newborn babies”.
James undertook postdoctoral work analysing trial results and his involvement with SurePulse really began when the work switched “from data analysis to business planning and fundraising. Working with academic colleagues, we put together a business plan and presented it to the university and our commercial partner, Tioga”.
Derby based Tioga are critical part of the SurePulse story as their expertise in electronics manufacture and device assembly has been a key factor in the successful development of the company’s products. James also cites “a large Innovate UK late stage biomedical catalyst grant” as being “key to initiating development once the business was established”.

Looking ahead to the future James says that SurePulse’s “first product has been CE marked and is on the market” and that the company feels it is “building traction, whilst developing its next generation product”. He reflects that “entry into the market has given us even deeper insight into the customer, and we’re really excited about the prospects this development will bring, both for the company and for new born babies across the world”